Introduction: Signal transducer and activator of transcription 3 (STAT3) has been shown to be critical to the survival of acute myeloid leukemia (AML) cells. It has been previously shown to mediate oxidative phosphorylation (OXPHOS) in acute myeloid leukemia bulk and stem cells via its transcriptional regulation of the amino acid transporter SLC1A5 and thereby glutamine uptake to AML cells (Amaya et al. Blood 2022). Further, STAT3 has been shown to regulate the transcription of important mitochondrial genes such as MCL-1 (Shastri et al. JCI 2018). Although STAT3 has been previously shown to also translocate to the mitochondria of myeloid malignant cells, its role in the mitochondria is less well defined in this cell population. In this study, we sought to understand the role of mitochondrial STAT3 in AML bulk leukemia and leukemia stem cell population.
Methods: Human primary AML samples and MOLM-13 cells were utilized for experiments. To assess STAT3 localization to the mitochondria, mitochondrial fractions were isolated followed by Western Blot analysis. Flow cytometry-based imaging (ImageStream) was also utilized to quantify localization of STAT3 to the mitochondria. Mitochondrial mass was assessed using flow cytometry based MitoTracker assays. Metabolomic assays were performed by mass spectrometry at the University of Colorado metabolomics core. Viability assays were performed with flow cytometry using Annexin V and Dapi stains. Patient derived xenograft models were performed as previously described (Amaya et al. Blood 2022) in NSG-S mouse with Busulfan conditioning.
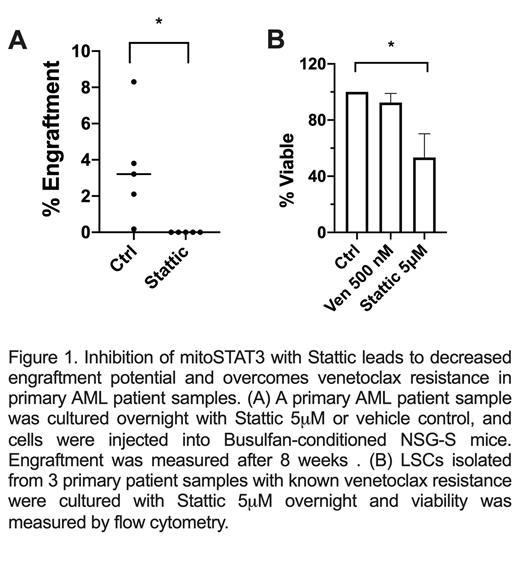
Results: In this study, we show that primary human AML cells have constitutive STAT3 activation via phosphorylation of S727, which has been shown to signal mitochondrial localization. Further, we show that STAT3 is localized in the mitochondria of leukemia cells in primary patient samples and the cell line MOLM-13. Using genetic (siRNA) and pharmacologic (Stattic) inhibition of STAT3, we show a decrease in pSTAT3 at S727, with subsequent decreased translocation of STAT3 to the mitochondria. This leads to mitochondrial dysfunction including decreased TCA cycle metabolites, as well as decreased intracellular ATP upon STAT3 inhibition. STAT3 inhibition also leads to decreased mitochondrial mass suggesting autophagy upon mitochondrial STAT3 (mitoSTAT3) inhibition. To assess whether mitoSTAT3 is functionally important, we performed viability assays in MOLM-13, as well as leukemia stem cells isolated from primary AML cells. Inhibition of mitochondrial STAT3 leads to decreased viability in MOLM-13 and primary AML cells, and reduced engraftment in PDX mouse models using primary AML samples (Figure 1A), suggesting a decrease in leukemia stem cell potential. Interestingly, STAT3 inhibition with Stattic is able to overcome venetoclax resistance for primary AML patient samples in vitro (Figure 1B).
Conclusions: In this study, we show mitoSTAT3 plays a key role in mitochondrial function of AML cells. mitoSTAT3 inhibition leads to mitochondrial dysfunction, a decrease in mitochondrial mass and eventually cell death. We show that Stattic is an effective inhibitor of mitoSTAT3 in AML, leading to cell death and decreased leukemia stem cell function. Further studies are needed to assess the mechanistic function of STAT3 in the mitochondria.
Disclosures
No relevant conflicts of interest to declare.